Surface Enhanced Raman Spectroscopy
Nanostructured Ag Triangle Films
Nanostructured silver films are fabricated using a technique known as Nanosphere Lithography (NSL). A monolayer of nanospheres, polystyrene in our experiments, serve as a mask for the thermal evaporation of silver.& Upon removal of the nanosphere mask a film of silver triangles remains.
Experimental details:
A monolayer of polystyrene spheres are is formed on a cleaned (Piranha:RCA) glass cover slip by slowly withdrawing it from a 1% aqueous suspension of nanospheres (Interfacial Dynamics, Alkjdadl, OR). The spheres order in a hexagonal closed packed structure in micron to tens of micron sized regions. Silver is then evaporated onto the sample and the spheres removed by sonication in ethanol. Triangles of silver are formed where silver deposited on the glass slide through the interstitial regions of the hexagonal closed packed spheres. Before examining the surface enhancing properties of these silver films, ~ 50 mL of Rhodamine 6G (1 mM in Methanol) is spun-cast onto the sample.
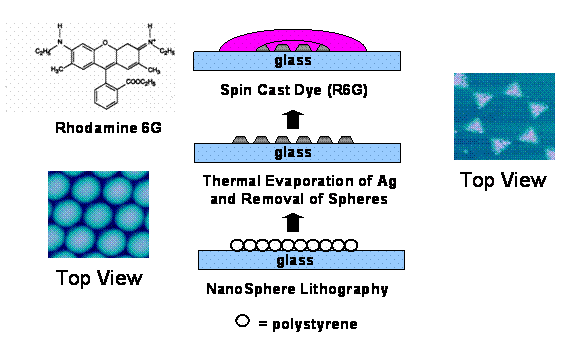
The size of the triangles (b) depends on the diameter (D) of spheres used to make the mask and can be roughly calculated from the following equation.

Characterization of Ag Triangle Films:
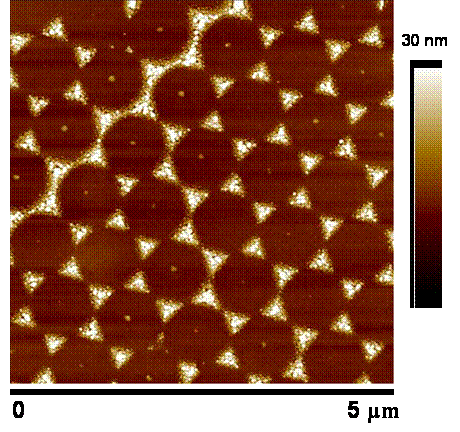
Both sphere masks and Ag triangle films are characterized by atomic force microscopy (AFM). The image below is a 5 x 5 ?m AFM image showing Ag triangles 30 nm tall.
Selected References:
- H. W. Deckman and J. H. Dunsmuir, Applied Physics Letters 41, 377-379 (1982).
- J. C. Hulteen and R. P. Vanduyne, Abstracts of Papers of the American Chemical Society 210, 25-Phys (1995).
- J. C. Hulteen and R. P. Vanduyne, Journal of Vacuum Science & Technology a-Vacuum Surfaces and Films 13, 1553-1558 (1995).
- R. P. VanDuyne, Abstracts of Papers of the American Chemical Society 213, 290-Phys (1997).
- F. Burmeister, C. Schafle, B. Keilhofer, C. Bechinger, J. Boneberg, and P. Leiderer, Chemical Engineering & Technology 21, 761-763 (1998).
- J. C. Hulteen, D. A. Treichel, M. T. Smith, M. L. Duval, T. R. Jensen, and R. P. Van Duyne, Journal of Physical Chemistry B 103, 3854-3863 (1999).
- T. R. Jensen, M. L. Duval, K. L. Kelly, A. A. Lazarides, G. C. Schatz, and R. P. Van Duyne, Journal of Physical Chemistry B 103, 9846-9853 (1999).
- T. R. Jensen, G. C. Schatz, and R. P. Van Duyne, Journal of Physical Chemistry B 103, 2394-2401 (1999).
- A. J. Haes, C. L. Haynes, A. D. McFarland, and R. P. Van Duyne, Abstracts of Papers of the American Chemical Society 222, U106-U106 (2001).
- C. L. Haynes, A. D. McFarland, M. T. Smith, J. C. Hulteen, and R. P. Van Duyne, Journal of Physical Chemistry B 106, 1898-1902 (2002).
- R. P. Van Duyne, Abstracts of Papers of the American Chemical Society 223, U63-U63 (2002).
- W. Kandulski, A. Kosiorek, J. Rybczynski, D. Bauman, and M. Giersig, Acta Physica Polonica A 104, 495-502 (2003).
- S. Reculusa and S. Ravaine, Chemistry of Materials 15, 598-605 (2003).
- A. D. Ormonde, E. C. M. Hicks, J. Castillo, and R. P. Van Duyne, Langmuir 20, 6927-6931 (2004).
- J. P. Schmidt, S. E. Cross, and S. K. Buratto, Journal of Chemical Physics 121, 10657-10659 (2004).
- A. J. Haes, J. Zhao, S. L. Zou, C. S. Own, L. D. Marks, G. C. Schatz, and R. P. Van Duyne, Journal of Physical Chemistry B 109, 11158-11162 (2005).
- F. Jarai-Szabo, S. Astilean, and Z. Neda, Chemical Physics Letters 408, 241-246 (2005).
- B. J. Y. Tan, C. H. Sow, T. S. Koh, K. C. Chin, A. T. S. Wee, and C. K. Ong, Journal of Physical Chemistry B 109, 11100-11109 (2005).
- Y. Yamaguchi, M. Ishikawa, Y. Maruyama, and M. Futamata, Journal of the Korean Physical Society 47, S56-S62 (2005).
